|
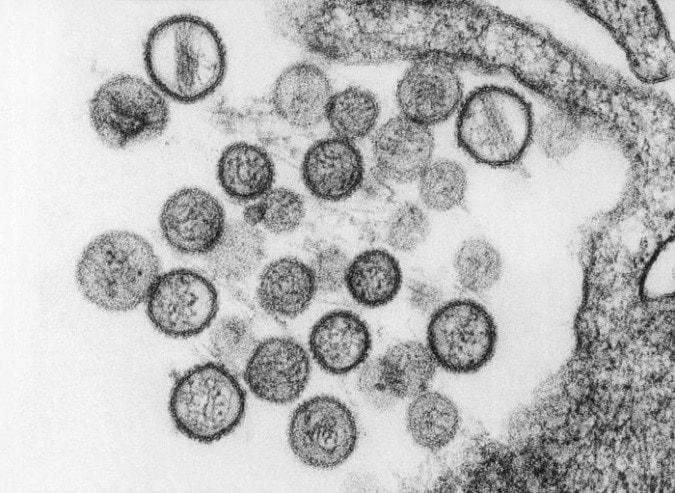
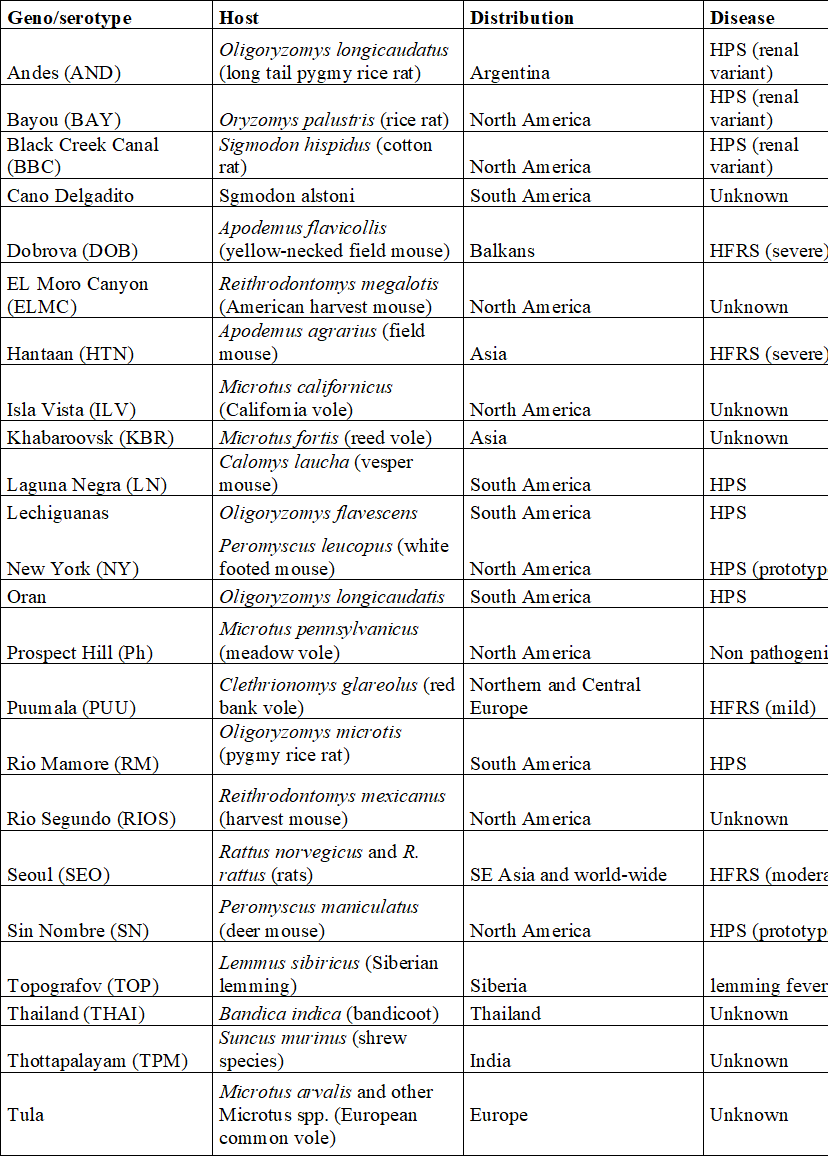
by Houssem Djeridi What are hantaviruses?Hantaviruses are a group of viruses that may be carried by some rodents. Some hantaviruses can cause a rare but deadly disease called hantavirus pulmonary syndrome (HPS). the related illness Hemorrhagic Fever with Renal Syndrome (HFRS) are rare infections caused by viruses belonging to the Bunyaviridae family. Viruses causing HPS are found in the Americas while those causing HFRS exist worldwide. Hantaviruses (genus Hantavirus, family Bunyaviridae) are a group of antigenically distinct RNA viruses carried in rodents and insectivores (shrews and moles). Each hantavirus is endemic in one, or at most, a few specific rodent or insectivore hosts, to which it is well adapted. At least 20 hantaviruses have been identified but estimates of the exact number of viruses vary. Newly identified hantaviruses are often named for the location where the virus is found; however, some of these viruses are later reclassified. Which rodents can cause humans to get Hantavirus ?Not all rodents carry hantavirus and there is usually no way to tell when a rodent has the virus. So, it is wise to avoid all contact with rodents when possible. Rodents, insectivores and bats known reservoir hosts for hantaviruses include rodents, insectivores (e.g., shrews and moles) and bats. Each virus is thought to be adapted to one or a few species, but spillover rodent, insectivore and bat hosts may not be unusual. Members of the mouse genus Apodemus carry Hantaan, Amur-Soochong and Dobrava-Belgrade viruses. Norway rats (Rattus norvegicus) are important reservoir hosts for Seoul virus; however, this virus has also been found in other species of rats including R. rattus (black rats), R. flavipectus, R. losea and R. nitidus. Bandicoot rats (Bandicota indica) carry Thailand virus, and bank voles (Myodes glareolus) are the reservoir hosts for Puumala virus. Tula virus has been found in several species of voles in the genus Microtus, in the water vole Arvicola amphibius and in the steppe lemming (Lagurus lagurus). Deer mice (Peromyscus maniculatus) carry Sin Nombre virus,while Black Creek Canal and Muleshoe viruses have been found in cotton rats (Sigmodon hispidus). Andes virus and its variants occur in rodents belonging to the South American mousegenera Akodon and Necromys and the rice rat genus Oligoryzomys. Laguna Negra virus has been detected in the vesper mice Calomys laucha and Calomys callidus, while Rio Mamore infects members of Oligoryzomys. Transmission
SymptomsHemorrhagic Fever with Renal Syndrome HFRS
Hantavirus Pulmonary Syndrome HP
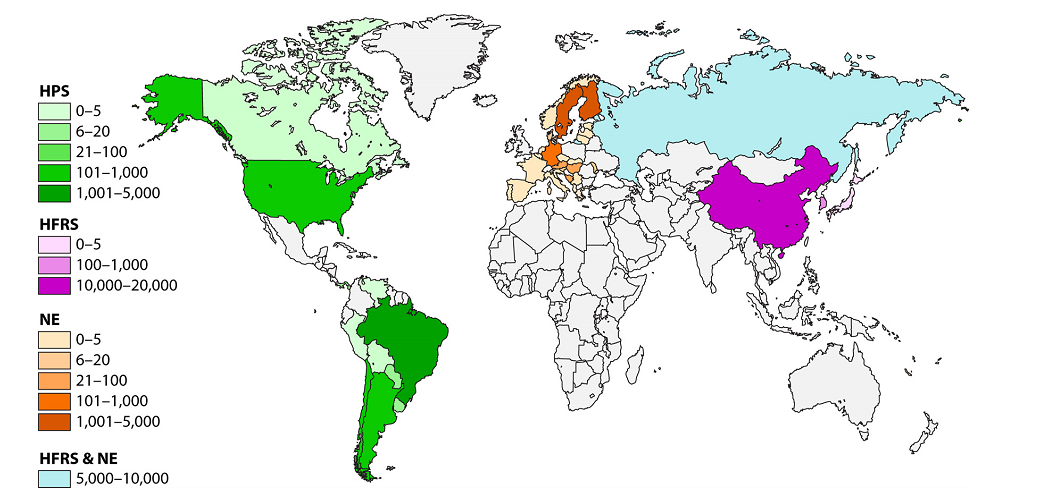
Geographic DistributionHantaviruses occur worldwide, but the distribution of each virus is limited by the geographic range of its reservoir host(s). Species known to be hantavirus carriers may or may not be infected in a given region. The hantaviruses in the Eastern Hemisphere are usually associated with HFRS. Seoul virus and Tula virus can be found in both Europe and Asia, while Dobrava-Belgrade and Puumala viruses circulate in Europe, and Hantaan, Amur-Shoochong, Gou, Thailand and Thottapalayam viruses occur in Asia. There are reports of Puumala or Puumala-like viruses in rodents in Asia. Sangassou, Bowe and Uluguru viruses occur in Africa. There is currently no evidence for hantavirus-associated disease in Australia, although seropositive rodents have been reported. Table: Hantavirus types, their hosts, Distribution and disease associationsThe most important hantaviruses causing HFRS are Hantaan virus, Puumala virus, Dobrava virus and Seoul virus. Other hantaviruses such as Amur virus are also associated with this disease. HFRS includes several diseases that formerly had other names, including Korean hemorrhagic fever and epidemic hemorrhagic fever. ”Nephropathia epidemica (NE)” is sometimes used for a mild form of HFRS, which is often caused by Puumala virus or Saaremaa virus. Hantavirus pulmonary syndrome is caused by a number of hantaviruses in North and South America. In the United States and Canada, the Sin Nombre virus (with its Monongahela and New York variants) is responsible for most cases. HPS can also result from infection by the Muleshoe, Bayou, and Black Creek Canal viruses, as well as other named or unnamed hantaviruses. In South and Central America, Andes virus and its variants are important causes of HPS; Choclo, Castelo Dos Sonhos, Juquitiba, Bermejo, Maciel and other hantaviruses can also cause this syndrome. Some hantaviruses have yet been not been linked to human disease, either because they are not pathogenic for humans or because their rodent hosts are unlikely to pass the virus to humans. Figure: Geographic distribution of approximate hantaviral disease incidence by country per year Incubation PeriodThe incubation period for HFRS can range from approximately one to 6 weeks, while incubation periods of 1-7 weeks have been reported in HPS. Many cases of HFRS and HPS seem to become apparent in about 2-3 weeks. Prevention • Keep mice and rats out of your home. • Clean up mouse and rat urine, droppings, and nesting materials with a disinfectant or a mixture of bleach and water. Hantaviruses are susceptible to many disinfectants including 1% sodium hypochlorite, 70% ethanol, 1-5% peracetic acid, A 10% sodium hypochlorite solution has been recommended for heavily soiled areas. Viruses in solution can be inactivated by heating to 56°C (133°F) for at least 15 minutes. Dried viruses were reported to be inactivated by 2 hours at 56°C. Treatment There is no specific treatment, cure, or vaccine for hantavirus infection. If infected individuals are recognized early and receive medical care in an intensive care unit, they may do better. In intensive care, patients are intubated and given oxygen therapy to help them through the period of severe respiratory distress. The earlier the patient is brought in to intensive care, the better. If a patient is experiencing full respiratory distress, it is less likely that the treatment will be effective. Diagnostic Tests HFRS and HPS are often diagnosed by serology. Antibody titers can usually be detected by the time the clinical signs develop, or soon afterward. Specific IgM or a rise in the IgG titer is diagnostic. ELISAs, immunochromatographic tests and immunofluorescent antibody tests (IFA) are the most commonly used serological tests, but other assays, including immunoblotting and virus neutralization, may also be available. Virus neutralization can distinguish serological reactions to different rodent-borne viruses, but the requirement for live virus limits its use. Antibodies to the hantaviruses carried in insectivores and bats may not be detected with the currently used serological tests. Clinical cases can also be diagnosed by detecting antigens in tissues with immunohistochemistry, or viral RNA in blood, saliva and tissues with RT-PCR. Some PCR tests use species-specific primers, but tests that can detect multiple hantaviruses have also been published. Nucleic acids may not be found in some patients by the time the symptoms develop. Conversely, one study detected Andes virus nucleic acids in some household contacts before they developed symptoms or became seropositive. Virus isolation can also be used for a definitive diagnosis; however, this is uncommon, due to the risks associated with culturing these viruses. In addition, some hantaviruses have never been successfully isolated in cell culture. Isolated hantaviruses can be identified by virus neutralization. Morbidity and Mortality Hantavirus outbreaks are often associated with increased rodent populations or environmental factors that promote human exposure to rodents. Clinical cases are reported to be seasonal in a number of areas. For instance, HPS is more common in late spring and early summer in the U.S., while HFRS occurs more frequently in winter and spring in China. Worldwide, approximately 150,000 to 200,000 people are estimated to be hospitalized with HFRS each year. Most of these cases occur in Asia, although several thousand illnesses are reported each year in Europe and Russia. Many of the clinical cases in Europe are caused by Puumala virus and are mild. HPS is also relatively common in some parts of South America; however, it seems to be infrequent in North America, with approximately 11-50 cases/ year reported in the U.S., and 0-13 cases/ year in Canada. The severity of HFRS and HPS varies with the causative virus and the availability and quality of healthcare. Improved diagnosis and supportive treatments have decreased the case fatality rates for some illnesses, compared to historical reports. The case fatality rate for HFRS ranges from < 0.5% or < 1% in nephropathia epidemica caused by Puumala virus or the Saaremaa strain of Dobrava-Belgrade virus, respectively, to 12% in some other Dobrava-Belgrade virus infections. Amur virus infections are also reported to be severe. Currently, the case fatality rate is reported to be approximately 1-2% for HFRS caused by Seoul virus and 5% for cases caused by Hantaan virus. Rates as high as 10-15% were reported for Hantaan virus in the past. HPS is frequently life-threatening, with case fatality rates estimated to range from 25% to 40% for most viruses. Approximately half of all cases are reported to be fatal in parts of Brazil where Araquara and Paranoá viruses are found, which suggests that these variants might be particularly virulent. Conversely, Laguna Negra virus and Choclo virus seem to cause less severe illnesses, with reported case fatality rates of 15% and 10%, respectively. Conclusion The emergence of zoonotic pathogens remains one of the great unsolved mysteries in biology. In the past century, numerous pathogens have emerged and reemerged, with an estimated frequency of one new pathogen every 18 months. Of these new pathogens, the majority have zoonotic origins in wildlife, and many have been RNA viruses such as the hantaviruses, severe acute respiratory syndrome coronavirus (SARS CoV), Ebola virus, Nipah virus, Hendra virus, West Nile virus, and, most recently, the 2009 H1N1 swine flu that initially emerged in Mexico. These and many other viruses harbored in vertebrates and invertebrates will continue to provide potential sources of new infections of humans and domestic or wild animals. There are many principles that have been learned from past epidemics and pandemics. Fundamentally, however, there remain gaps in our understanding of the ecology and natural history of zoonotic viruses; how they are maintained in their reservoirs; the processes and mechanisms that lead to transmission, host switching, recombination, and reassortment; and molecular events that lead to the transfer and adaptation to a new host and posttransfer adaptation. Gentaur offers many products for the study and diagnosis of hantavirus, check our products here:
0 Comments
Leave a Reply. |
Genprice Inc. USA
(718) 513-2983
[email protected]
Gentaur SRL Italy
02 36 00 65 93
[email protected]
Gentaur GmbH Germany
+49 0241 40 08 90 86
[email protected]
Gentaur Ltd. UK
020 3393 8531
[email protected]
Gentaur Spain
0911876558
[email protected]
Gentaur Sarl France
01 43 25 01 50
[email protected]
Gentaur BV Netherlands
0208-080893
[email protected]
Gentaur BVBA Belgium
0032 265 09 23
[email protected]
Gentaur Sp. z o.o. Poland
058 710 33 44
[email protected]
(718) 513-2983
[email protected]
Gentaur SRL Italy
02 36 00 65 93
[email protected]
Gentaur GmbH Germany
+49 0241 40 08 90 86
[email protected]
Gentaur Ltd. UK
020 3393 8531
[email protected]
Gentaur Spain
0911876558
[email protected]
Gentaur Sarl France
01 43 25 01 50
[email protected]
Gentaur BV Netherlands
0208-080893
[email protected]
Gentaur BVBA Belgium
0032 265 09 23
[email protected]
Gentaur Sp. z o.o. Poland
058 710 33 44
[email protected]